!
Polycrystalline Silicon Preparation by Silane Method
1. Introduction
Silicon is a widely used element in the semiconductor industry, and the preparation of polycrystalline silicon is an important process due to the large-scale development of solar cells. Polycrystalline silicon is prepared by the silane method, which is a low-temperature chemical vapor deposition process that combines the characteristics of high efficiency and low energy consumption as compared to physical vapor deposition methods. In this paper, the Silane method is introduced and discussed, its main reaction process, gas source selection and process parameters optimization.
2. Silane Method
The crystal structure of silicon films is determined by the deposition temperature and growth time. When the deposition temperature is low, the average grain size becomes smaller, forming a polycrystalline structure, while when the deposition temperature is increased, the grain growth of the deposited film increases and a single crystal structure is obtained. The advantage of the silane method is that the process can be carried out at a low temperature from 300 to 450 ˚C, which is much lower than the 1000 ˚C target temperature of the hot wire method.
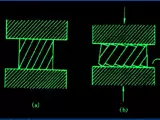
The silane-based method for preparing polycrystalline silicon is a chemical vapor deposition (CVD) method, also known as the SiH4-based method. Figure 1 shows the principle of the silane method and its main reactions, where SiCl4 and SiH4 are used as sources of silicon and the reaction is carried out in a hydrogen-rich atmosphere. The SiCl4 molecules first hydrolyze with water to form H2SiO3, which further decomposes to SiH4 and HCl. The SiH4 then reacts with hydrogen to produce silicon and water. The reaction process of silane also produces chlorine and hydrogen chlorides, which can have an effect on the film growth.
< img src=Figure1.jpg alt=Figure1 />
Figure 1. Principle of Silane Method and Its Main Reactions
3. Gas Source Selection
The reaction process of the silane method is affected by the type of gas source and its flow rate. The choice of gas source depends on the needs of the silicon film, such as size and morphology. SiH4 is the key gas for producing silicon films, and for a high deposition rate, H2 is usually selected as the carrier gas. Other gas sources such as methane, propane, carbon dioxide and nitrogen can also be used as carrier gases. They can effectively reduce the growth rate of the silicon films and lower the deposition temperature.
4. Process Parameters Optimization
The deposition process of the silane method is affected by process parameters such as pressure, temperature, flow rate and film thickness. Pressure is an important parameter for controlling the deposition rate of the film and is affected by the choice of gas source. The temperature is also an important parameter and is affected by the pressure. The flow rate of the carrier gas and SiH4 also affects the deposition rate of the film, so it is important to make sure that the flow rate is properly controlled.
5. Conclusion
Polycrystalline silicon can be prepared by the silane method, which is a low temperature chemical vapor deposition process. The reaction process of the silane method is affected by the type of gas source and its flow rate, and process parameters such as pressure, temperature, flow rate and film thickness must be optimized for optimal results. The advantages of the silane method include its ability to produce high-quality films at a low deposition temperature.